chapter 5 electrochemistry class 12 chemistry textbook solution
4. Answer the following :
vii. What is a cell constant ? What are its units? How is it determined experimentally?
Answer:-
Cell Constant:
The conductivity of an electrolytic solution is given by Equation:
k = 1 / (R * l * a)
For a given cell, the ratio of separation (l) between the two electrodes divided by the area of cross-section (a) of the electrode is called the cell constant. Thus,
Cell Constant = l / a
The SI unit of cell constant is m-1, which is conveniently expressed in cm-1. Equation (5.14) then becomes
k = cell constant / R
Determination of cell constant
- The determination of the cell constant involves the use of 1 M, 0.1 M, or 0.01 M KCl solutions. The conductivity values for KCl solutions at various temperatures are readily available in tables.
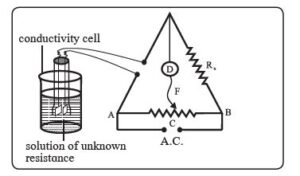
- The resistance of the KCl solution is measured using a Wheatstone bridge, as illustrated in the figure.
- AB represents a uniform wire, while R_x signifies a variable known resistance placed in one arm of the Wheatstone bridge. The other arm of the bridge contains the conductivity cell, which holds the KCl solution with an unknown resistance.
- D serves as the current detector, and F denotes the sliding contact that moves along AB. A.C. represents the source of alternating current.
- The sliding contact is adjusted along AB until no current flows through the system, as indicated by the absence of deflection on the detector D. This position is referred to as the null point and is achieved at C.
- According to the Wheatstone bridge principle:
\( \frac{{R_{\text{{solution}}}}}{{l_{\text{{AC}}}}} = \frac{{R_x}}{{l_{\text{{BC}}}}} \)
Hence, \( R_{\text{{solution}}} = \frac{{l_{\text{{AC}}}}}{{l_{\text{{BC}}}}} R_x \)
The resistance of the KCl solution can be determined by measuring the lengths AC and BC, along with knowledge of R_x.
The cell constant is defined as:
Cell constant = k_KCl × “R_solution”
Since the conductivity of the KCl solution is well-established, the cell constant can be computed accordingly.
chapter 5 elctrochemistry textbook solution page 118
