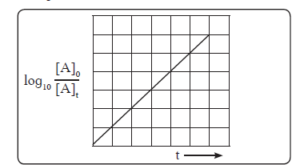
xi. How will you represent first order reactions graphically.
Answer:- i. The differential rate law for the first order reaction A→ P is
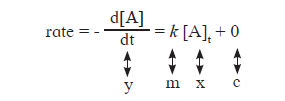
The equation is of the form y = mx + c. A plot of rate versus concentration [A]t is a straight line passing through origin. This is shown in Fig. 6.3. The slope of straight line = k.
![Variation of rate with [A]](https://cracknta.com/wp-content/uploads/2023/09/Variation-of-rate-with-A-300x186.png)
ii. From Eq. the integrated rate law is
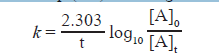

![A plot showing log10 [A]t vs time t](https://cracknta.com/wp-content/uploads/2023/09/A-plot-showing-log10-At-vs-time-t-300x149.png)