12. ALDEHYDES, KETONES AND CARBOXYLIC ACIDS
4. Answer the following
iv. Write reaction showing conversion of p- bromoisopropyl benzene into p-Isopropyl benzoic acid ( 3 steps).
Answer:-
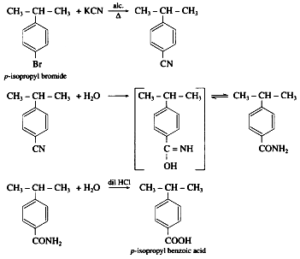
Step 1: Bromination of p-Isopropylbenzene The first step is to brominate p-isopropylbenzene to introduce a bromine atom (Br) onto the benzene ring. This reaction can be carried out using bromine (Br2) in the presence of a Lewis acid catalyst, such as iron (Fe):
Reaction: p-Isopropylbenzene + Br2/Fe → p-Bromoisopropylbenzene
Step 2: Oxidation of p-Bromoisopropylbenzene to p-Isopropylbenzaldehyde The second step involves the oxidation of p-bromoisopropylbenzene to form p-isopropylbenzaldehyde. This can be accomplished using a mild oxidizing agent like sodium dichromate (Na2Cr2O7) in an acidic medium (e.g., sulfuric acid, H2SO4):
Reaction: p-Bromoisopropylbenzene + Na2Cr2O7/H2SO4 → p-Isopropylbenzaldehyde + NaBr + Cr2(SO4)3 + H2O
Step 3: Oxidation of p-Isopropylbenzaldehyde to p-Isopropylbenzoic Acid The final step is the conversion of p-isopropylbenzaldehyde into p-isopropylbenzoic acid. This can be achieved through further oxidation using a stronger oxidizing agent like potassium permanganate (KMnO4) in an acidic medium:
Reaction: p-Isopropylbenzaldehyde + KMnO4/H+ → p-Isopropylbenzoic acid + MnO2 + H2O
After these three steps, you will have successfully converted p-bromoisopropylbenzene into p-isopropylbenzoic acid. Please note that proper safety precautions and laboratory techniques should be followed when conducting these reactions, especially when working with chemicals like bromine and strong oxidizing agents.
12. ALDEHYDES, KETONES AND CARBOXYLIC ACIDS page 281
Question bank with Solution
Text book Solution
NCERT Solutions
NCERT Solutions for Class 12 Maths
NCERT Solutions for Class 12 Physics
NCERT Solutions for Class 12 Chemistry
NCERT Solutions for Class 12 Biology
NCERT Solutions for Class 11 Maths
NCERT Solutions for Class 11 Physics
NCERT Solutions for Class 11 Chemistry
NCERT Solutions for Class 11 Biology
NCERT Solutions for Class 10 Maths
NCERT Solutions for Class 10 Science
Board/University Study Material
Question Paper Solutions
CBSE Previous Year Question Paper With Solution for Class 12 Arts
CBSE Previous Year Question Paper With Solution for Class 12 Commerce
CBSE Previous Year Question Paper With Solution for Class 12 Science
CBSE Previous Year Question Paper With Solution for Class 10
Maharashtra State Board Previous Year Question Paper With Solution for Class 12 Arts
Maharashtra State Board Previous Year Question Paper With Solution for Class 12 Commerce
Maharashtra State Board Previous Year Question Paper With Solution for Class 12 Science
Maharashtra State Board Previous Year Questio n Paper With Solution for Class 10
CISCE ICSE / ISC Board Previous Year Question Paper With Solution for Class 12 Arts
CISCE ICSE / ISC Board Previous Year Question Paper With Solution for Class 12 Commerce
CISCE ICSE / ISC Board Previous Year Question Paper With Solution for Class 12 Science
CISCE ICSE / ISC Board Previous Year Question Paper With Solution for Class 10
