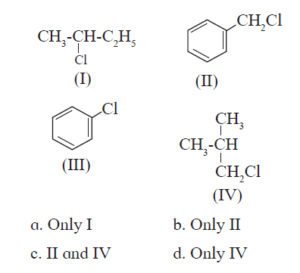
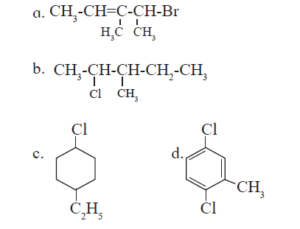
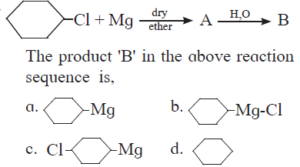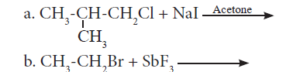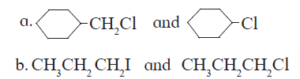10. HALOGEN DERIVATIVES Textbook Solution
i. The correct order of increasing reactivity of C-X bond towards nucleophile in the following
compounds is
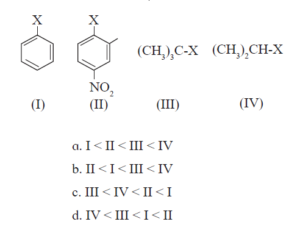
a. I < II < III < IV
b. II < I < III < IV
c. III < IV < II < I
d. IV < III < I < II
I – CH2 – CH = CH2
CH3 – CH2 – CH2I
\[\begin{array}{cc} \ce{CH3-CH-CH3}\\ |\\ \ce{I}\end{array}\] \[\begin{array}{cc} \ce{CH3-CH-CH2}\\ \phantom{…..}|\phantom{…..}|\\ \phantom{…….}\ce{I}\phantom{….}\ce{OH} \end{array}\]iv. The best method for preparation of alkyl fluorides is
a. Finkelstein reaction
b. Swartz reaction
c. Free radical fluorination
d. Sandmeyer’s reaction
v. Identify the chiral molecule from the following.
a. 1-Bromobutane
b. 1,1- Dibromobutane
c. 2,3- Dibromobutane
d. 2-Bromobutane
vi. An alkyl chloride on Wurtz reaction gives 2,2,5,5-tetramethylhexane. The same alkyl chloride on reduction with zinc-copper couple in alchol give hydrocarbon with molecular formula C5H12. What is the structure of alkyl chloride
\[\begin{array}{cc}
\ce{CH3}\\
|\phantom{...}\\
\ce{CH3-C-CH2Cl}\\
|\phantom{...}\\
\ce{CH3}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3\phantom{..}}\\
|\phantom{.....}\\
\ce{CH3-C-CH2CH3}\\
|\phantom{.....}\\
\ce{Cl\phantom{....}}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3-CH2-CH-Cl}\\
\phantom{......}|\\
\ce{\phantom{.........}CH3}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3-CH-CH-CHCl}\\
|\phantom{.....}|\phantom{...}\\
\ce{\phantom{..}CH3} \ce{\phantom{..}CH3\phantom{..}}
\end{array}\]
vii. Butanenitrile may be prepared by heating
a. propanol with KCN
b. butanol with KCN
c. n-butyl chloride with KCN
d. n-propyl chloride with KCN
viii. Choose the compound from the following that will react fastest by SN1 mechanism.
a. 1-iodobutane
b. 1-iodopropane
c. 2-iodo-2 methylbutane
d. 2-iodo-3-methylbutane
x. Which of the following is used as source of dichlorocarbene
a. tetrachloromethane
b. chloroform
c. iodoform
d. DDT
iii. Identify chiral molecule/s from the following.
a.
\[\begin{array}{cc}\ce{CH3 - CH - CH2 - CH3}\\|\phantom{.........}\\\ce{OH\phantom{.......}}\end{array}\]
b.
\[\begin{array}{cc}\ce{CH3 - CH2 - CH - CH2 - CH3}\\
|\\\ce{Br}\end{array}\]
c. CH3 - CH - CH2 - CH2Br
d.
\[\begin{array}{cc}\ce{CH3 - CH - CH2 - CH3}\\
|\phantom{.........}\\\ce{CH3\phantom{......}}\end{array}\]
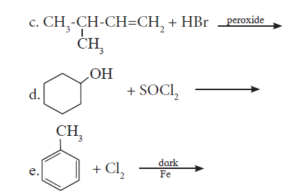
1. \[\ce{CH3 – CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
2. \[\begin{array}{cc}\ce{CH3 – CH = CH2 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{…………………….}\\
\ce{OH\phantom{…………………..}}\end{array}\]
3. \[\begin{array}{cc}\ce{CH3\phantom{…..}}\\
|\phantom{…….}\\
\ce{CH3 – C – CH2 – Cl ->[Na/dry ether]}\\
|\phantom{…….}\\
\ce{CH3\phantom{…..}}
\end{array}\]
vi. Name the reagent used to bring about the following conversions.
a. Bromoethane to ethoxyethane
b.1-Chloropropane to 1 nitropropane
c. Ethyl bromide to ethyl isocyanide
d. Chlorobenzene to biphenyl
vii. Arrange the following in the increase order of boiling points
a. 1-Bromopropane
b. 2- Bromopropane
c. 1- Bromobutane
d. 1-Bromo-2-methylpropane
Match the following
| Column I | Column II |
| \[\begin{array}{cc}\ce{CH3CH – CH3}\\|\phantom{….}\\ \ce{X\phantom{….}}\end{array}\] | vinyl halide |
| CH2 = CH – CH2X | alkyl halide |
| CH2 = CH – X | allyl halide |
| benzyl halide | |
| aryl halide |
i. Haloarenes are less reactive than halo alkanes.
ii. Alkyl halides though polar are immiscible with water.
iii. Reactions involving Grignard reagent must be carried out under anhydrous condition.
iv. Alkyl halides are generally not prepared by free radical halogenation of alkanes.
4. Distinguish between – SN1 and SN2 mechanism of substitution reaction ?
5. Explain – Optical isomerism in 2-chlorobutane.
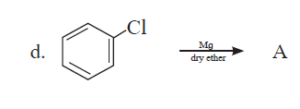
6. Convert the following.
i. Propene to propan-1-ol
ii. Benzyl alcohol to benzyl cyanide
iii. Ethanol to propane nitrile
iv. But-1-ene to n-butyl iodide
v. 2-Chloropropane to propan-1-ol
vi. tert-Butyl bromide to isobutyl bromide
vii. p-Nitrochlorobenzene to p-nitrophenol
viii. Propene to 1-nitropropane
i. HCl is added to a hydrocarbon ‘A’ (C4H8) to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ (C4H10O). Identify ‘A’ , ‘B’ and ‘C’.
iii. Observe the following and answer the questions given below.
a. Name the type of halogen derivative
b. Comment on the bond length of C-X bond in it
c. Can react by SN1 mechanism? Justify your answer.